Why A2 Milk’s Recall May Not Justify Its 12% Decline

Key Takeaways
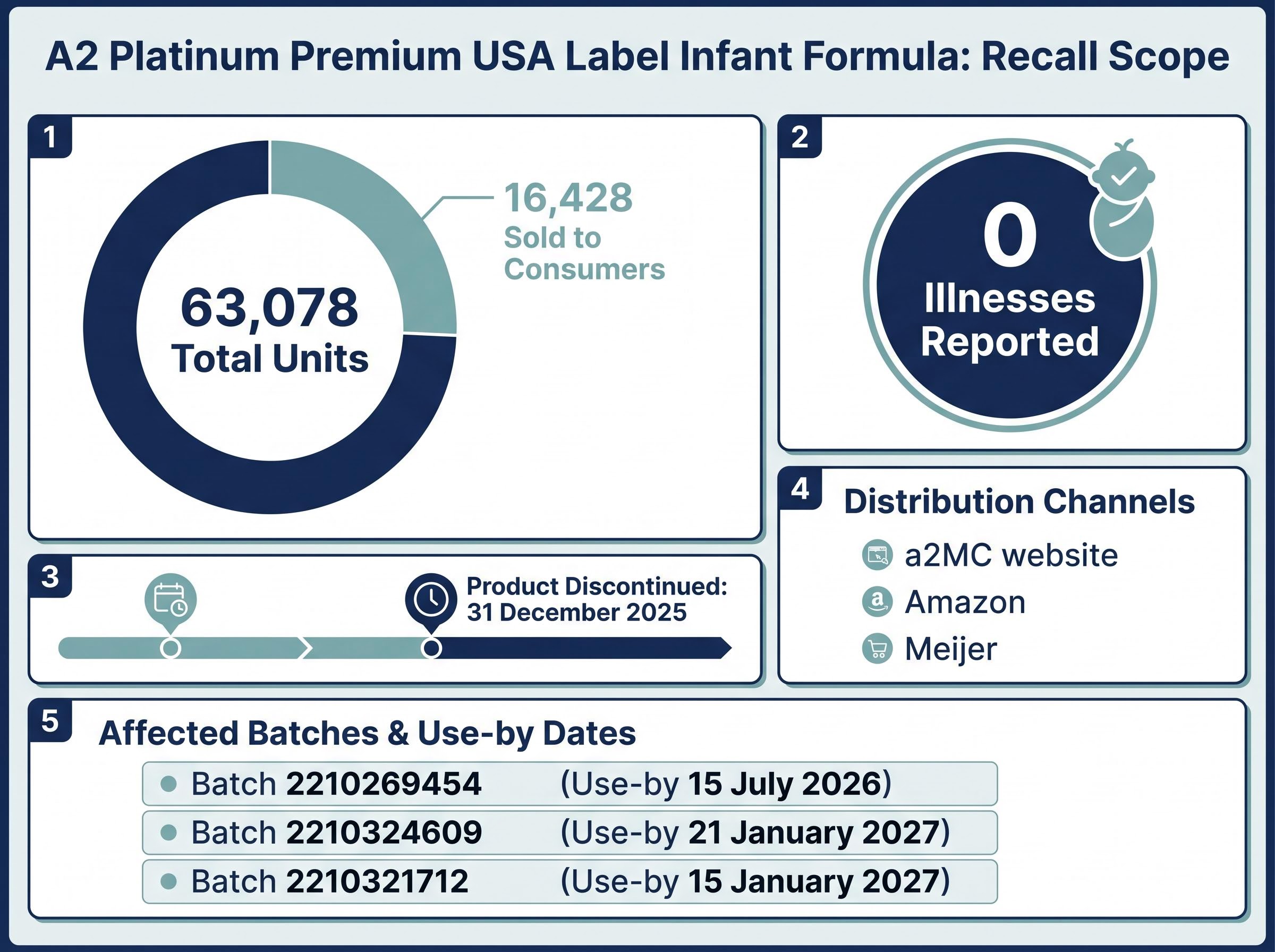
- The A2 Milk product recall covers 63,078 tins of a discontinued US infant formula line, with zero reported illnesses and narrow distribution limited to a2MC's website, Amazon, and Meijer stores.
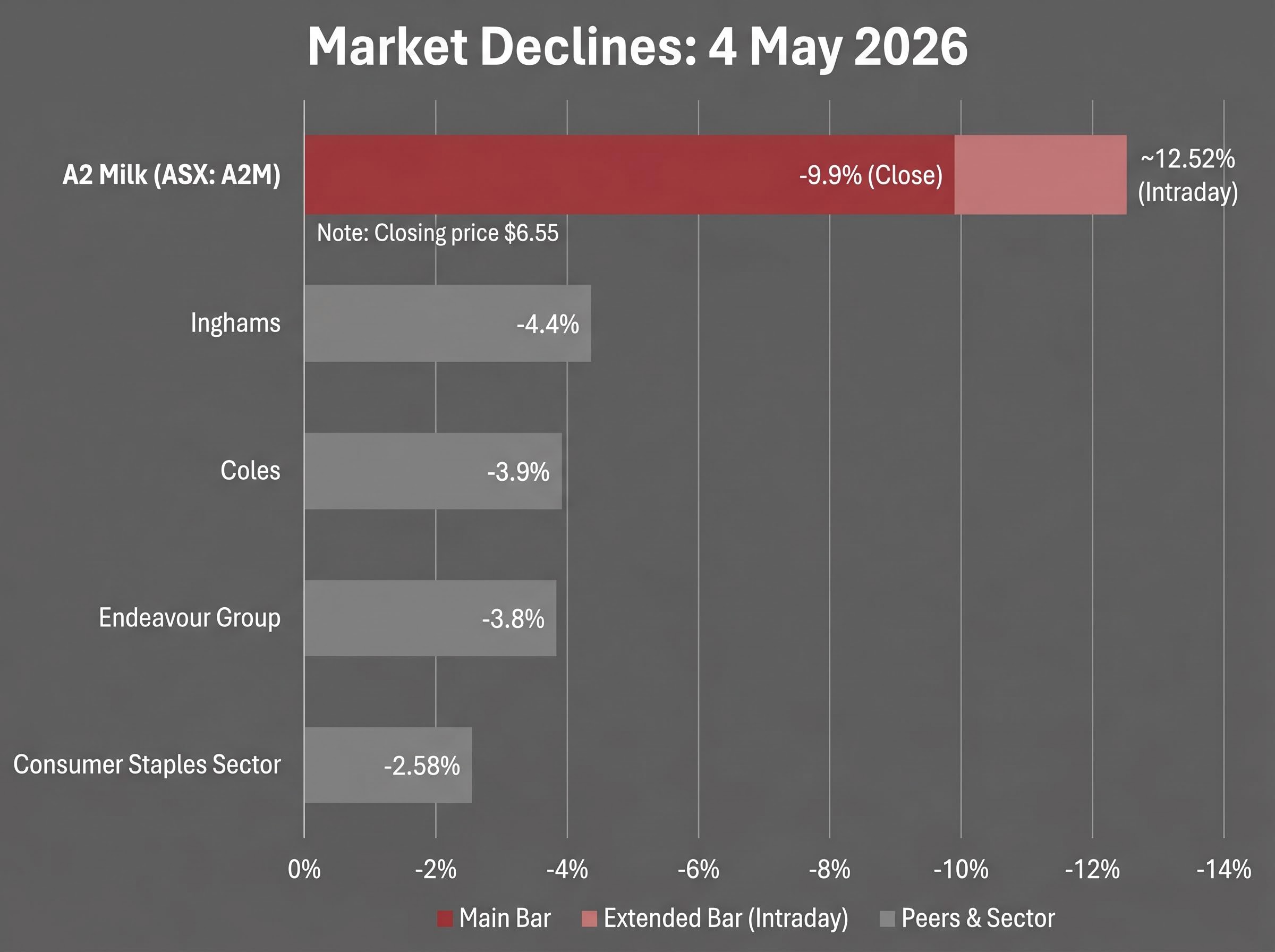
- A2M shares fell 9.9% at close and reached a 52-week low on 4 May 2026, but Consumer Staples peers without recall exposure also fell 3-4% on the same session, suggesting the recall accounts for only part of the decline.
- The analyst consensus 12-month price target for A2M stands at $8.98 AUD against the $6.55 close on 4 May 2026, implying approximately 37% upside at consensus.
- Similar precautionary actions by Nestle, Danone, and Lactalis in 2025-2026 suggest an industry-wide testing regime rather than an isolated quality failure unique to a2 Milk.
- The recall compounds existing investment case pressures including an FY26 EBITDA margin guidance cut to 14.0%-14.5%, with FDA classification outcome and broker response notes serving as the key near-term catalysts to monitor.
A company voluntarily pulls 63,078 tins of infant formula from US shelves, reports zero illnesses, and watches its shares fall more than 12% to a 52-week low on the same day. The numbers do not obviously add up, and that gap is worth examining closely.
On 4 May 2026, The a2 Milk Company (ASX: A2M) became the session’s largest percentage decliner in the ASX 300, falling 9.9% to $6.55 on the back of a voluntary US recall announced two days earlier. The product at the centre of the recall had already been discontinued at the end of 2025. Consumer Staples was the worst-performing sector on the day, adding a macro layer of selling pressure to a stock already absorbing company-specific news.
What follows unpacks the exact scope of the A2 Milk product recall, explains what cereulide contamination means for a vulnerable consumer population, tests whether the direct financial exposure justifies the market’s reaction, and provides a framework for assessing what product recall events historically mean for consumer goods share prices.
What the recall actually covers, and what it does not
The recall announced on 2 May 2026 involves three batches of a2 Platinum Premium USA label infant formula (0-12 months, 31.7oz tins). The total unit count is 63,078, of which approximately 16,428 were sold to consumers. Distribution was narrow: the product was available only through a2MC’s website, Amazon, and Meijer stores under Operation Fly Formula, the US government-facilitated import programme introduced during the 2022 infant formula shortage.
The three affected batches:
- Batch 2210269454, use-by 15 July 2026
- Batch 2210324609, use-by 21 January 2027
- Batch 2210321712, use-by 15 January 2027
| Batch Number | Use-By Date | Distribution Channel |
|---|---|---|
| 2210269454 | 15 July 2026 | a2MC website, Amazon, Meijer |
| 2210324609 | 21 January 2027 | a2MC website, Amazon, Meijer |
| 2210321712 | 15 January 2027 | a2MC website, Amazon, Meijer |
The product line was discontinued on 31 December 2025 following the expiration of US importation rights. No ongoing revenue stream exists to be disrupted. The US Food and Drug Administration (FDA) is involved, though no formal Class I, II, or III classification has been assigned; the recall is voluntary and precautionary.
The FDA voluntary recall announcement confirms the precautionary framing of the action, noting no illnesses in connection with the affected product and documenting the narrow distribution footprint across the three named channels.
No illnesses have been reported in connection with the affected products.
The recall’s defined edges matter for investors. A recall of a discontinued product with limited distribution is categorically different from a live product line withdrawal.
When big ASX news breaks, our subscribers know first
Cereulide contamination and why infant formula is the highest-stakes context
Cereulide is a heat-stable toxin produced by certain strains of the bacterium Bacillus cereus. The heat stability is what matters here: normal warming or preparation of powdered formula cannot destroy the toxin once present.
The FSANZ guidance on cereulide in infant formula details the heat-stable properties of the toxin and the specific health effects observed in infants, including the rapid onset window and the elevated severity risk tied to lower body weight in this consumer cohort.
Health effects in affected individuals include:
- Nausea, vomiting, abdominal cramps, and diarrhoea
- Symptom onset typically within 1-6 hours of consumption
- In severe cases, dehydration and liver failure
Why infant formula raises the stakes
Infants are a specifically vulnerable population. Their lower body weight means smaller quantities of the toxin can produce more severe effects, and their inability to communicate symptoms raises the risk of delayed detection.
The contamination was identified through additional testing prompted by guidance from New Zealand food authorities. Synlait Milk, the manufacturer, confirmed compliance with production standards at the time of manufacture. This framing is relevant: what occurred appears to be a post-production detection rather than a manufacturing failure, suggesting improved testing protocols identified contamination that may previously have gone undetected.
For investors, that distinction matters. The recall could not reasonably have been avoided once the contamination was detected, which bears on any assessment of management’s handling of the event.
The share price reaction in the context of the broader session
The sell-off on 4 May 2026 was severe, but it did not occur in isolation. Consumer Staples was the worst-performing sector on the session, falling 2.58% to 11,853.1. The broader S&P/ASX 200 declined 0.38% to 8,697.1, with an RBA rate decision expected on 5 May 2026 (a 25 basis point increase was forecast), creating an independently negative backdrop for consumer-facing equities.
| Company | Decline (%) | Reported Driver |
|---|---|---|
| A2 Milk (ASX: A2M) | 9.9% (close); ~12.52% intraday | US infant formula recall |
| Coles | 3.9% | Sector-wide selling |
| Inghams | 4.4% | Sector-wide selling |
| Endeavour Group | 3.8% | Sector-wide selling |
On the NZX, ATM fell 11.24% to $7.90, reaching a 52-week low.
A2M shares dropped 12.52% at the open before recovering to close at $6.36, a partial intraday recovery that itself reflects the market’s rapid reassessment of the recall’s narrow scope once analysts and investors had time to read the specifics of the affected batch and distribution channel.
The analyst consensus 12-month price target for A2M stands at $8.98 AUD, against the $6.55 close on 4 May 2026, implying approximately 37% upside at consensus.
Disentangling the layers is the analytical challenge. Consumer Staples names without any recall exposure fell 3-4% on the same session. That suggests perhaps 6-9 percentage points of A2M’s decline is attributable to the recall itself, with the remainder reflecting sector rotation and pre-RBA positioning. Neither explanation excuses the other, but the distinction shapes how an investor reads the implied market discount.
How the industry’s recall pattern changes the reputational calculus
A2 Milk is not the only infant formula producer to address Bacillus cereus or cereulide concerns in the current cycle.
- Nestlé addressed cereulide concerns in its own infant formula lines during 2025-2026
- Danone undertook precautionary actions related to Bacillus cereus contamination in the same period
- Lactalis conducted similar product safety measures in response to heightened testing standards
This pattern suggests an industry-wide elevation of testing and detection thresholds rather than an isolated quality failure at a2 Milk or its contract manufacturer.
Detection versus negligence: why the distinction matters for investors
There is a material difference between a company with a systemic manufacturing problem and a company whose product was flagged by improved testing protocols that are being applied across the sector. Synlait Milk’s confirmation that production standards were met at the time of manufacture supports the latter interpretation.
If the market is applying a company-specific quality discount to A2M when the recall reflects a sector-wide testing regime, that mispricing could partially correct as the broader industry context becomes better understood. The reputational risk remains real, but it is shared, not unique.
A framework for reading single-session recall declines as an investor signal
The question at the centre of any recall sell-off is whether the event represents a lasting impairment to future earnings or a one-time reputational shock that the market will reprice once specifics are absorbed.
Three variables historically determine whether recall-driven declines recover:
- Breadth of affected product. Is the recall contained to a specific, limited batch or does it affect an active, revenue-generating product line?
- Illness reports. Have consumers been harmed, or is the action precautionary?
- Systemic versus one-time. Does the recall point to an ongoing manufacturing failure, or was it a one-time batch detection?
Applying the framework to A2M specifically
On all three variables, the A2M recall sits closer to the recoverable end of the spectrum. The 63,078 affected units belong to a product line discontinued on 31 December 2025. Zero illnesses have been reported as of 4 May 2026. The contamination context is sector-wide, not company-specific.
One factor complicates the picture. Infant formula occupies a category where consumer and regulatory trust is rebuilt slowly. Parents choosing formula for newborns apply a higher standard of caution than consumers of adult food products. That reputational sensitivity could extend the discount period beyond what the framework’s factual inputs would otherwise suggest.
The shares closed at a 52-week low with an analyst consensus target of $8.98 AUD, but the gap between the two is not, on its own, a signal to act. It is a measure of the question the market is debating.
The next major ASX story will hit our subscribers first
Limited direct exposure, but the 52-week low warrants monitoring
The direct financial exposure is genuinely contained. The recalled product line was discontinued, the affected unit count is small relative to group operations, and no live revenue stream faces disruption. No parallel recall actions have been confirmed in Australia or New Zealand.
The recall compounds an investment case that was already under pressure before 4 May 2026: a prior FY26 guidance downgrade cut EBITDA margin expectations to 14.0%-14.5% from 15.5%-16.0%, with Synlait manufacturing backlogs and elevated cereulide testing requirements among the five converging supply constraints management identified in April.
The legitimate concerns sit elsewhere. Reputational sensitivity in the infant formula category is unusually high. No formal broker rating revisions had been issued as of the close on 4 May 2026. A stock reaching a 52-week low in a weak sector environment may attract further technical selling before fundamentals reassert.
For investors monitoring A2M at its 52-week low, our deep-dive into short interest data as an early warning system examines how institutional short sellers built positions in other ASX names weeks before significant price moves occurred, and explains the four-business-day publication lag that shapes how retail investors can practically use the signal.
A fully insurance-covered class action settlement reached in April 2026 removed one established legal overhang from the A2M investment case, meaning the recall now lands on a company that had recently cleared a significant liability concern rather than one carrying multiple unresolved legal exposures simultaneously.
The forward monitoring triggers are specific:
- Any emergence of illness reports linked to the affected batches
- Formal FDA classification of the recall (Class I would signal heightened severity)
- Broker target revisions in the days following the announcement
- Any indication that the contamination source affects other a2 Milk product lines
The gap between the $8.98 analyst consensus target and the $6.55 close implies approximately 37% upside at consensus, a spread wide enough to warrant close attention to whichever catalyst narrows it first.
This article is for informational purposes only and should not be considered financial advice. Investors should conduct their own research and consult with financial professionals before making investment decisions. Past performance does not guarantee future results.
The gap between headline risk and actual impairment rarely stays wide for long
The recall’s direct financial footprint is small. The product is discontinued. The industry context reduces the reputational uniqueness of the event. Those are the facts as they stand.
The countervailing reality is that infant formula is a category where consumer and regulatory trust is rebuilt slowly, even when evidence supports a precautionary rather than negligent framing. The FDA classification outcome, next-session volume patterns, and broker response notes will sharpen the picture materially in coming days.
As of 4 May 2026, zero illnesses have been reported. That remains the single most consequential variable in the forward outlook.
The framework built here applies not only to this recall, but to the next one in the sector. The distinction between headline risk and actual earnings impairment is where analytical value lives.
Frequently Asked Questions
What is the A2 Milk product recall about?
The recall involves 63,078 tins of a2 Platinum Premium USA label infant formula across three batches, voluntarily pulled from US shelves on 2 May 2026 due to possible cereulide contamination. The product line had already been discontinued on 31 December 2025, and no illnesses have been reported.
What is cereulide contamination and why does it matter for infant formula?
Cereulide is a heat-stable toxin produced by certain strains of Bacillus cereus that cannot be destroyed by normal formula preparation or warming. It poses a heightened risk to infants due to their lower body weight and inability to communicate symptoms, making early detection and precautionary recalls especially important in this product category.
Why did A2 Milk shares fall so sharply if the recalled product was already discontinued?
A2M shares fell approximately 9.9% at close and 12.52% intraday on 4 May 2026, reflecting a combination of recall-specific concern and broader Consumer Staples sector selling, with the sector itself down 2.58% on the same session. Analysts estimate roughly 6-9 percentage points of the decline were attributable to the recall, with the remainder linked to sector rotation and pre-RBA positioning.
Is the A2 Milk recall isolated or part of a broader infant formula industry pattern?
The recall appears to reflect an industry-wide elevation of testing and detection standards rather than a company-specific failure, with Nestle, Danone, and Lactalis all undertaking similar precautionary actions related to Bacillus cereus during 2025-2026. Synlait Milk, the manufacturer, confirmed that production standards were met at the time of manufacture.
What should investors monitor following the A2 Milk recall announcement?
Investors should watch for any emergence of illness reports linked to the affected batches, a formal FDA recall classification (particularly a Class I designation), broker target revisions in the days following the announcement, and any indication that contamination affects other a2 Milk product lines beyond the discontinued US label.