Lumos Diagnostics secures US$507k BARDA milestone after FebriDx CLIA waiver

Lumos secures US$0.5 million BARDA milestone payment following FebriDx CLIA waiver achievement
Lumos Diagnostics Holdings has received a milestone payment of US$0.5 million from the Biomedical Advanced Research and Development Authority (BARDA) following the achievement of US FDA CLIA waiver status for its FebriDx rapid diagnostic test. The payment brings total CLIA waiver study funding received from BARDA to US$2,984,571 under contract number 75A50124C00051, providing non-dilutive capital that extends the company’s capacity to advance clinical programmes without shareholder dilution.
The milestone payment validates regulatory progress achieved through the BARDA partnership, which has provided both funding and technical expertise throughout the CLIA waiver development process. CLIA waiver status represents the lowest regulatory complexity classification for diagnostic tests in the United States, enabling deployment across healthcare settings that previously could not access FebriDx under its former moderate-complexity designation.
CEO commentary on BARDA partnership value
Doug Ward
“BARDA’s assistance provides not only meaningful non-dilutive funding that extends our capacity to run critical studies, including our paediatric programme, but also brings technical expertise as we advance our diagnostic solutions toward broader clinical use.”
The BARDA contract supports multiple clinical development initiatives beyond the CLIA waiver achievement. Management has highlighted the paediatric programme as a key focus area enabled by the partnership’s funding structure, which removes capital constraints that typically limit early-stage diagnostic companies from pursuing parallel development pathways.
When big ASX news breaks, our subscribers know first
What is CLIA waiver and why does it matter for diagnostic companies?
CLIA waiver is a US FDA classification that permits diagnostic tests to be used in simple healthcare settings without laboratory infrastructure or specialised operator training. The Clinical Laboratory Improvement Amendments (CLIA) programme establishes quality standards for laboratory testing, with “waived” tests representing those deemed simple enough for use by non-laboratory personnel in point-of-care environments.
Tests classified as “moderate-complexity” — FebriDx’s previous designation — require certified laboratory facilities and trained operators, restricting deployment to hospital laboratories and dedicated diagnostic centres. CLIA waiver removes these barriers entirely, permitting use by general practitioners, nurses, and allied health professionals across primary care and retail health settings.
The CLIA waiver designation unlocks access to over 300,000 locations across the United States that hold a Certificate of Waiver. These locations include:
- Primary care physician offices
- Urgent care clinics
- Retail health and pharmacy clinics
- Community health centres
For diagnostic companies, CLIA waiver represents the difference between niche laboratory distribution and mass-market point-of-care deployment. Products restricted to moderate-complexity classification face structural limits on addressable market size due to the concentrated number of certified laboratory facilities compared to the distributed network of waived testing sites.
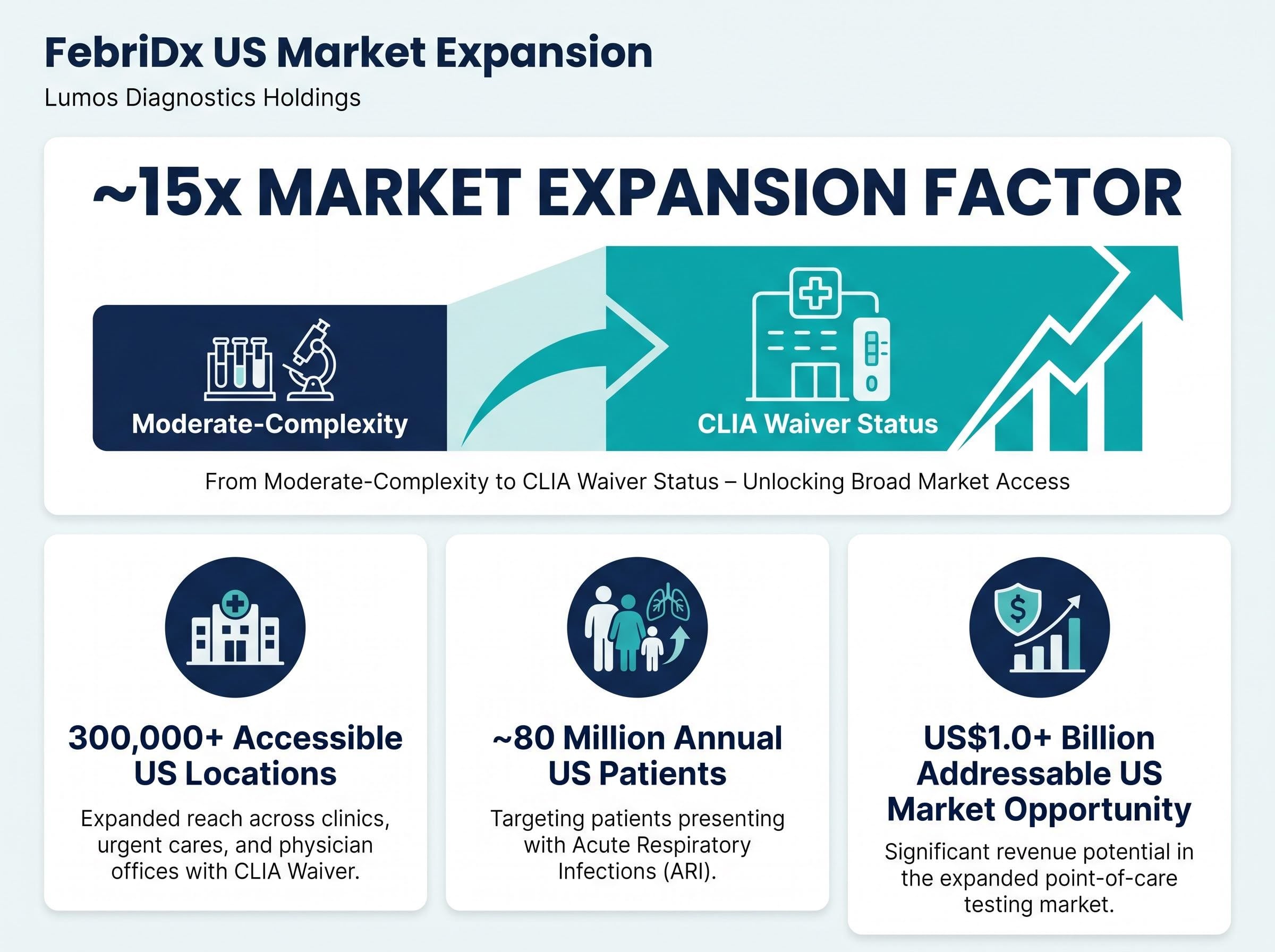
FebriDx market opportunity expands fifteen-fold following CLIA waiver
The CLIA waiver clearance expands FebriDx’s addressable market by approximately 15 times compared to its previous moderate-complexity classification. The company can now target approximately 80 million US patients per annum who present with acute respiratory infections at primary care and urgent care centres, representing a market opportunity exceeding US$1.0 billion annually.
| Metric | Value |
|---|---|
| US locations now accessible | 300,000+ |
| Annual patient reach (acute respiratory infections) | ~80 million |
| Addressable US market opportunity | US$1.0+ billion |
| Market expansion factor | ~15x |
FebriDx differentiates between bacterial and non-bacterial respiratory infections within 10 minutes, supporting antibiotic stewardship programmes by enabling clinicians to make informed prescribing decisions at the point of care. The test addresses a clinical workflow bottleneck where practitioners traditionally prescribe antibiotics empirically whilst awaiting laboratory culture results, contributing to antimicrobial resistance through unnecessary antibiotic use.
What FebriDx does and its clinical value
FebriDx is a rapid point-of-care test that assists healthcare professionals in distinguishing bacterial respiratory infections from viral or non-bacterial causes within 10 minutes. The test measures biomarkers associated with bacterial infection and systemic inflammation, providing clinicians with objective data to inform antibiotic prescribing decisions during patient consultations.
The clinical value proposition centres on antimicrobial stewardship, a global health priority focused on reducing unnecessary antibiotic prescriptions that drive drug-resistant bacterial infections. Traditional clinical practice often involves empirical antibiotic prescribing for respiratory infections due to the difficulty of rapidly determining bacterial versus viral causation without laboratory testing.
FebriDx shifts this paradigm by delivering actionable diagnostic information whilst the patient remains in the consultation room, potentially reducing antibiotic prescriptions for viral infections that do not benefit from antibacterial treatment. This positions the product within policy frameworks supporting antimicrobial stewardship, including initiatives by the US Centers for Disease Control and Prevention and the World Health Organisation.
The next major ASX story will hit our subscribers first
Next steps for Lumos commercialisation
With CLIA waiver clearance now secured, Lumos transitions from regulatory development to commercial deployment across the expanded US market. The company retains BARDA support for ongoing clinical programmes, including paediatric studies, whilst scaling distribution infrastructure to address over 300,000 waived testing locations.
The paediatric FebriDx milestone of US$720,000 was triggered earlier in 2026 under the same BARDA contract, confirming study progress toward potential clearance for children aged 2-12 years and demonstrating the sequential funding structure that underpins the paediatric programme.
The commercial pathway focuses on three key deployment channels:
- Continue BARDA-supported clinical programmes including paediatric study
- Scale US commercial distribution across 300,000+ waived settings
- Target primary care and urgent care channels representing 80 million annual patient interactions
Management has not disclosed specific commercialisation timelines or distribution partnership structures for the CLIA waiver market entry. The paediatric programme represents a clinical development priority, with BARDA funding assisting with ongoing clinical programmes including paediatric studies.
The CLIA waiver achievement removes the primary regulatory barrier to mass-market deployment, shifting execution risk from regulatory approval to commercial distribution and market adoption. Success in penetrating primary care and urgent care channels will determine whether Lumos can convert the expanded addressable market into material revenue generation.
Investors tracking how Lumos is converting the CLIA waiver into real deployment volume can find our detailed coverage of the WellStreet urgent care rollout, which examines the 43-site expansion across three US regions, the standing order protocol embedding FebriDx into standard clinical workflow, and the pathway to activating the full 163-location network.
Want the Next Healthcare Breakthrough in Your Inbox?
Join 20,000+ investors receiving FREE breaking ASX healthcare news delivered within minutes of release, complete with in-depth analysis. Click the “Free Alerts” button at StockWire X to get market-moving announcements the moment they break, with expert coverage already done for you.