A2 Milk Recalls 63,000 US Formula Tins as Shares Fall 12%

Key Takeaways
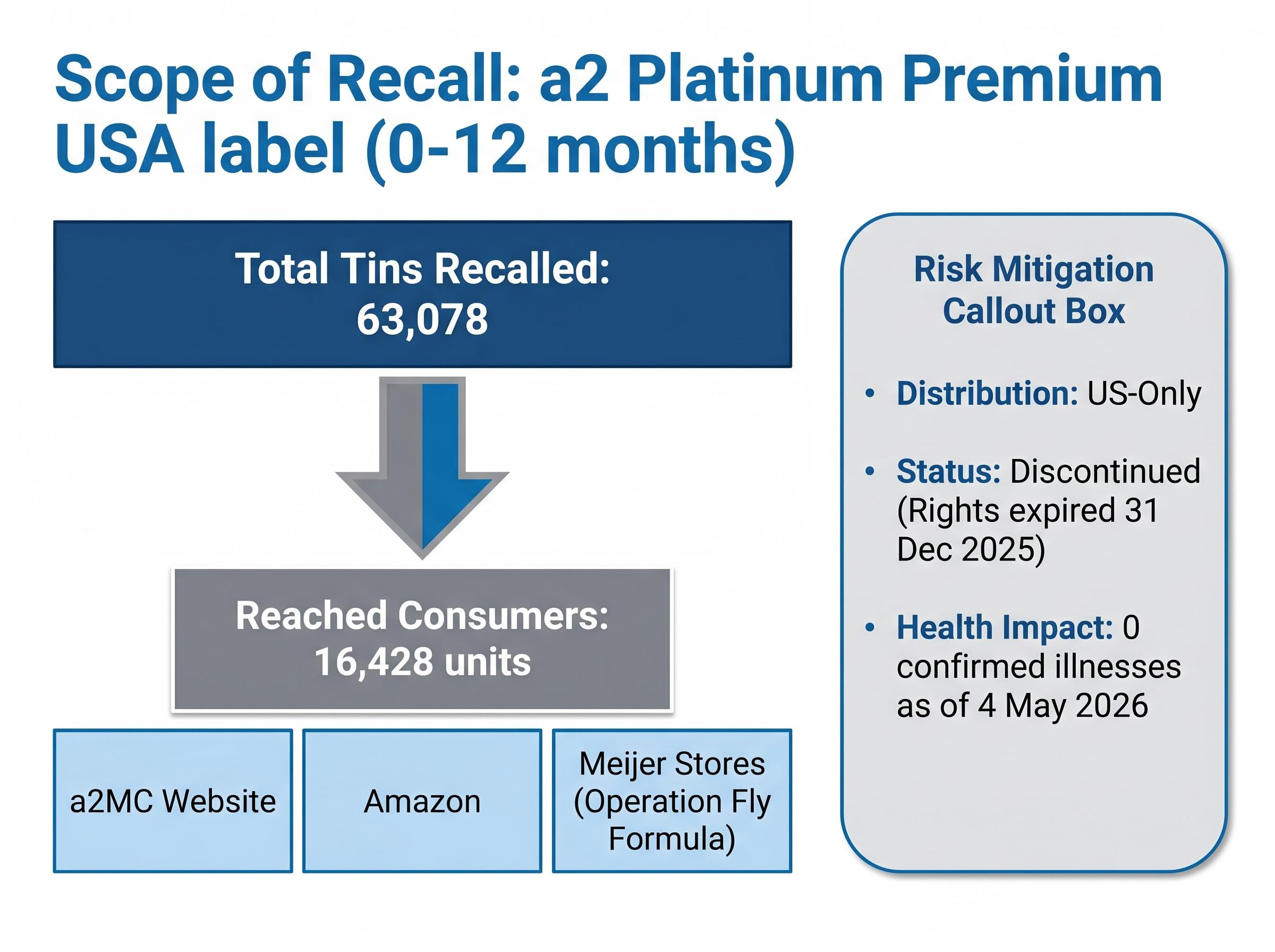
- A2 Milk voluntarily recalled approximately 63,078 tins of US infant formula after cereulide, a heat-stable bacterial toxin, was detected in an input ingredient, with no confirmed illnesses reported as of 4 May 2026.
- The recalled product was already discontinued, with US importation rights having expired on 31 December 2025, limiting the direct financial impact on A2M's ongoing revenue.
- A2M shares dropped 12.52% at the open before recovering to close at $6.36, reflecting the market's rapid reassessment of the recall's narrow scope.
- The recall is a US-only event with no impact on Australian or New Zealand consumers, and should not be conflated with a separate, unrelated Western Australia milk recall involving a different product and contaminant.
- The recall compounds an already-challenged investment case, with A2M down approximately 20.8% year-to-date before the announcement and carrying a prior FY26 guidance downgrade that cut EBITDA margin expectations to 14.0%-14.5%.
A2 Milk shares dropped sharply at Monday’s open after the company announced a voluntary recall of roughly 63,000 tins of infant formula sold in the United States, citing detection of cereulide, a bacterial toxin produced by certain strains of Bacillus cereus. The announcement landed during an already-weak session for the S&P/ASX 200, which was down 0.38% at midday on 4 May 2026, with a string of large-cap earnings disappointments weighing on broader sentiment. For A2M shareholders, the recall introduced a product safety dimension to a stock already under pressure, down approximately 20.8% year-to-date before the news broke. What follows covers the recall’s scope, what cereulide actually is and why it matters in infant formula, how the regulatory sequence unfolded, and what the intraday price action reveals about how markets process food safety shocks.
What A2 Milk actually recalled and who was affected
The recall covers three batches of a2 Platinum Premium USA label infant formula (0-12 months), totalling 63,078 tins. Of those, an estimated 16,428 units reached consumers through three distribution channels:
- a2MC’s own website
- Amazon
- Meijer stores, under Operation Fly Formula, the White House emergency initiative launched in 2022 to address domestic infant formula shortages
The product had already been discontinued before the recall was initiated. Importation rights expired on 31 December 2025, meaning no new stock was entering the US market.
No confirmed illnesses have been reported in connection with the recalled product as of 4 May 2026.
For Australian investors, the boundaries matter immediately. This is a US-only distribution event. No recalled product reached Australian or New Zealand consumers, and no concurrent cereulide-related recall has been issued by Food Standards Australia New Zealand (FSANZ). The direct consumer safety risk is confined to a narrow, already-discontinued US product line.
When big ASX news breaks, our subscribers know first
Cereulide explained: why this toxin is particularly dangerous in baby formula
Cereulide is not an ordinary bacterial contaminant. It is a toxin produced by certain strains of Bacillus cereus, and its defining characteristic is heat stability. Unlike many foodborne pathogens that are neutralised by cooking, cereulide survives heating and reheating. That property matters in infant formula, where preparation typically involves mixing powder with hot water.
The EFSA rapid risk assessment on cereulide in infant formula established acute reference doses and safety thresholds for the toxin, confirming that its heat stability is the primary property driving regulatory concern when contaminated powder is prepared with hot water.
The probable source of contamination was identified as an input ingredient rather than the finished product, suggesting the toxin entered the supply chain before the formula was manufactured.
The contamination-to-risk pathway follows a clear sequence:
- Cereulide forms in the contaminated ingredient before or during production
- The toxin survives the heat applied during formula preparation
- The infant ingests the prepared formula
- Symptoms develop, typically within 30 minutes to six hours
In healthy adults, the symptom profile is primarily gastrointestinal: nausea and vomiting that typically self-resolve within 24 hours.
Why the developing immune system changes the risk calculation
Infants face a different risk profile. Their developing immune systems and smaller body mass mean the primary danger is not the toxin itself causing severe acute harm, but a secondary complication: dehydration from repeated vomiting, which may require medical intervention. This is why regulatory and company responses were immediate despite the absence of confirmed illness reports. In the infant formula category, the threshold for action sits well below confirmed harm.
How the recall came about: the regulatory chain from New Zealand to the FDA
The recall was initiated voluntarily by A2 Milk following additional testing prompted by a regulatory update from New Zealand’s Ministry for Primary Industries (MPI). That sequence matters. This was a proactive, regulator-prompted response, not a reaction to reported illness or a compelled enforcement action.
The US Food and Drug Administration (FDA) is aware of and collaborating on the recall. No independent enforcement action or formal punitive response has been confirmed as of 4 May 2026.
The FDA’s collaborative posture, rather than adversarial, suggests the agency views the company’s voluntary action as appropriate to the identified risk.
Australian consumers face no direct exposure, but a separate, unrelated recall should not be conflated with this event:
- US infant formula recall: a2 Platinum Premium USA label, cereulide contamination, voluntary, US-only
- Western Australia milk recall: a2 Light Milk 2L, Listeria monocytogenes contamination, distinct product and distinct contaminant
The two events share a brand name and little else.
Reading the intraday price action: what the A2M chart told investors on 4 May
A2M opened sharply lower on the recall announcement. The verified intraday decline reached 12.52% on 4 May 2026, with the stock closing at $6.36.
Then the recovery began. By 12:53 pm AEST, A2M had gained from its opening price, trading at a higher level. The shape of the session told a story of rapid repricing as investors assessed the actual scope of the event.
| Time | A2M Price | Change from Open |
|---|---|---|
| Market open (sell-off low) | ~$6.14 | -12.52% (intraday decline) |
| 12:53 pm AEST | $6.59 | +7.15% from open |
| Market close | $6.36 | Session close |
The intraday recovery from the opening low reflects the market’s real-time reassessment as details of the recall’s limited scope became clear.
The recovery makes more sense when the moderating factors are assembled: US-only distribution, an already-discontinued product, no confirmed illnesses, and a voluntary recall initiated by the company rather than compelled by a regulator. Each detail, as it was absorbed, narrowed the perceived risk.
The broader session context also matters. The ASX 200 was down 0.38% at midday, with NAB’s credit impairment surge and Endeavour Group’s slowing sales adding to sentiment headwinds. A2M entered the day already approximately 20.8% lower year-to-date, meaning some of the initial sell-off likely reflected reflexive risk-off positioning on a stock already under pressure, rather than a fresh fundamental reassessment.
ASX market breadth had already been deteriorating in the week leading into the session, with 22 index constituents hitting 52-week lows in the week ending 1 May 2026 even as the headline ASX 200 fell only 0.65%, a divergence that amplified the reflexive risk-off pressure on individual stocks already under stress.
The next major ASX story will hit our subscribers first
The bigger picture for A2M investors after the recall clears
The direct financial exposure is narrow. The recalled product was already discontinued, distributed only in the US under a government emergency programme that has since ended, and carries no ongoing revenue contribution. The earnings impact, on available evidence, is limited.
Direct financial impact factors:
- Recalled product discontinued (importation rights expired 31 December 2025)
- US-only distribution with no ongoing revenue from this product line
- No confirmed consumer harm, reducing the probability of material litigation exposure
Reputational risk factors:
- The infant formula category is brand-sensitive globally; a recall in any market carries symbolic weight
- A2 Milk’s primary revenue markets, Australia and China, are not directly implicated by this US-only event
- Ingredient-sourced contamination (rather than post-production error) raises questions about supplier oversight that may require monitoring
The stock’s pre-existing 20.8% year-to-date decline complicates the picture. Investors assessing whether the recall materially changes the fundamental outlook should note that no analyst commentary or broker target changes specific to this event had been published by midday on 4 May 2026. The full institutional reassessment remains ahead, and updates from brokers in coming days will carry more weight than the headline-driven intraday move.
The recall lands on top of an already-challenged investment case: A2M’s FY26 guidance downgrade in April 2026 cut revenue growth expectations and slashed EBITDA margin guidance to 14.0%-14.5%, with China supply chain disruptions pushing revenue recognition into FY27 and cash conversion falling to approximately 50% from a prior forecast of 80%.
A considered response, not a crisis, but watch the brand
On the available evidence, A2 Milk’s recall was voluntary, regulator-prompted, limited to a discontinued US product, and has produced no confirmed illness reports. This is a well-managed product safety event rather than a systemic production failure.
The legitimate watch points remain. The infant formula category is unforgiving of brand damage. A2M’s share price was already under pressure before the announcement. The cereulide contamination of an ingredient, rather than a post-production error, raises questions about supplier oversight that will require monitoring in future reporting periods.
The intraday recovery suggests the market reached a similar conclusion by midday, but the absence of broker commentary means the full reassessment is still ahead. Investors may wish to monitor analyst updates in coming sessions before drawing firm conclusions about forward positioning.
For investors wanting to track the manufacturing dependency risk in more depth, our full explainer on Synlait’s recovery roadmap covers the three-stage stabilise-simplify-scale strategy, the North Island asset sale, and the new customer agreements that will shape A2M’s supply chain options as Pokeno production ramps in H1 FY27.
This article is for informational purposes only and should not be considered financial advice. Investors should conduct their own research and consult with financial professionals before making investment decisions.
—
Frequently Asked Questions
What is the A2 Milk infant formula recall about?
A2 Milk voluntarily recalled approximately 63,078 tins of its a2 Platinum Premium USA label infant formula (0-12 months) due to the detection of cereulide, a heat-stable bacterial toxin produced by certain strains of Bacillus cereus. The recall is limited to US distribution and covers three specific batches, with no confirmed illnesses reported as of 4 May 2026.
Is the A2 Milk recall affecting Australian or New Zealand consumers?
No. The recall is confined to US distribution only, and no recalled product reached Australian or New Zealand consumers. Food Standards Australia New Zealand (FSANZ) has not issued any cereulide-related recall for A2 Milk products.
What is cereulide and why is it dangerous in infant formula?
Cereulide is a toxin produced by certain strains of Bacillus cereus that is heat-stable, meaning it survives the hot water used to prepare infant formula. Infants are at heightened risk because repeated vomiting can cause dehydration serious enough to require medical intervention, even though healthy adults typically recover within 24 hours.
How did A2M shares react to the recall announcement on 4 May 2026?
A2M shares fell as much as 12.52% at the open on 4 May 2026, hitting an intraday low of approximately $6.14, before recovering to $6.59 by 12:53 pm AEST and closing the session at $6.36. The partial recovery reflected the market repricing the event once the limited scope became clearer.
What should investors watch after the A2 Milk recall?
Investors should monitor broker and analyst updates in coming sessions, as no target price changes specific to this event had been published by midday on 4 May 2026. Longer-term watch points include A2 Milk's supplier oversight processes given the contamination originated in an input ingredient, and any brand impact in the company's primary revenue markets of Australia and China.