Neurizon Wins Ethics Nod for Oral Liquid Trial Supporting ALS Disease Progression

Neurizon Therapeutics (ASX: NUZ) has received approval from the Bellberry Human Research Ethics Committee (HREC) for a Phase 1 formulation study of its NUZ-001 oral liquid formulation. The approval marks a clinical development milestone for NUZ-001 in amyotrophic lateral sclerosis (ALS) treatment, with the oral liquid formulation developed specifically to address swallowing difficulties experienced by ALS patients as their disease progresses.
Neurizon secures ethics approval for Phase 1 oral liquid formulation study
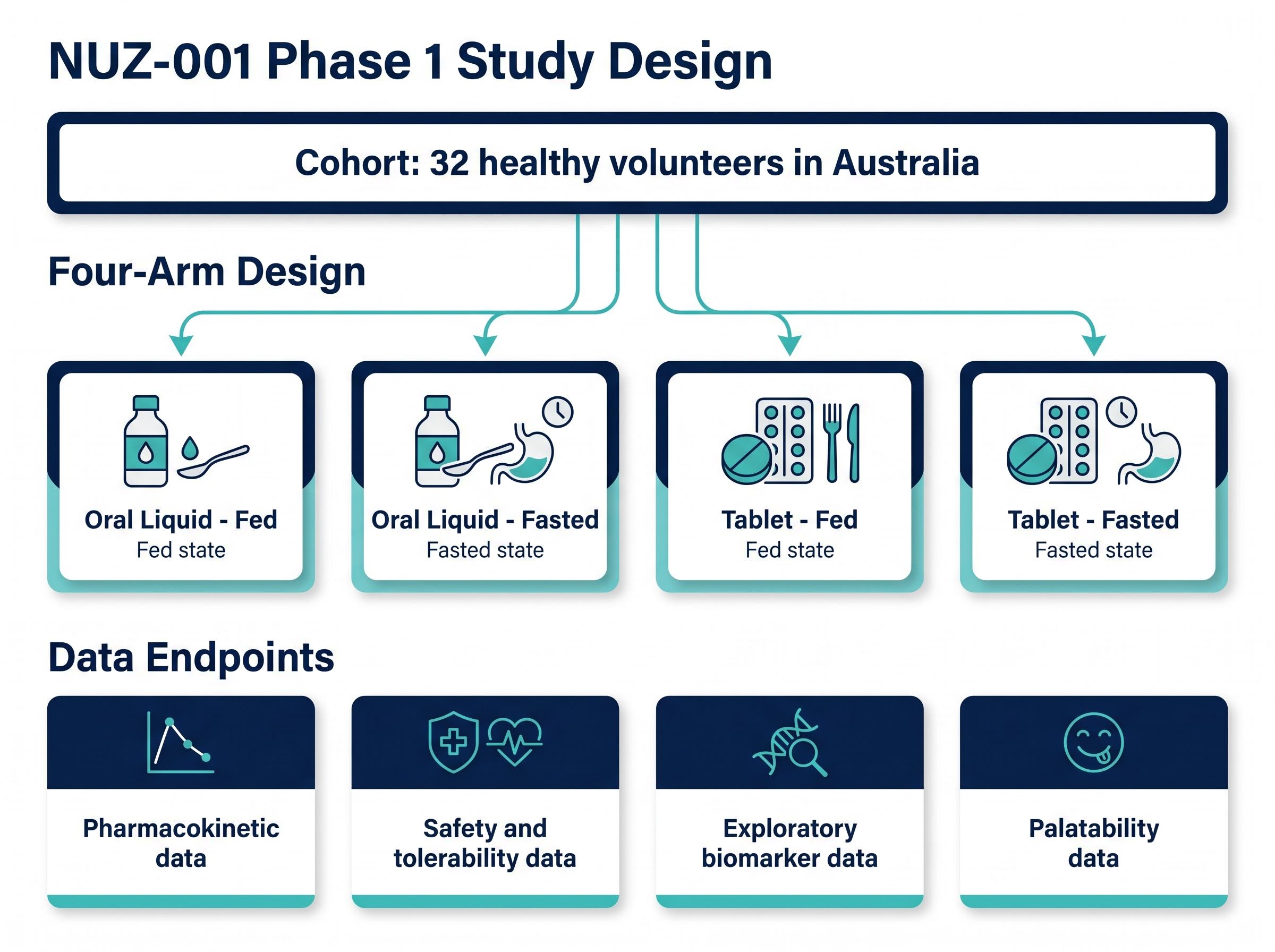
The HREC approval allows Neurizon to proceed with a Phase 1 clinical trial evaluating the oral liquid formulation of NUZ-001, representing execution on the company’s formulation development strategy announced in June 2025. The trial will enrol 32 healthy volunteers in Australia in a randomised, four-arm design evaluating both oral liquid and tablet formulations under fed and fasted conditions.
The oral liquid formulation was developed to improve treatment accessibility, administration flexibility, and continuity of care across different stages of ALS progression. For investors, ethics approval represents a critical gating milestone that de-risks the development timeline and confirms management is delivering on stated development plans.
ALS is associated with progressive impairment in speech and swallowing function (dysphagia). As the disease advances, patients may struggle to take tablet medications, potentially forcing treatment discontinuation due to administration challenges rather than clinical factors. An oral liquid formulation allows patients to continue therapy as their condition progresses, supporting longer-term treatment across disease stages.
When big ASX news breaks, our subscribers know first
Why oral liquid formulations matter in ALS treatment
ALS causes progressive muscle weakness, including the muscles used for swallowing. This degenerative condition means that swallowing function declines over time, creating practical barriers to tablet-based treatments. Patients in advanced disease stages may become unable to safely swallow solid medications, despite potential clinical benefit from continued treatment.
The oral liquid formulation addresses this challenge by providing an alternative administration route that remains viable as dysphagia worsens. This formulation flexibility can extend the duration patients remain on therapy, rather than being forced to discontinue due to mechanical difficulties unrelated to drug efficacy or tolerability.
For investors, this translates to potential commercial advantage. Formulation flexibility expands the addressable patient population to include those with moderate to severe swallowing impairment, and extends treatment duration across disease progression. If NUZ-001 reaches market, these factors could support broader adoption and longer patient retention compared to tablet-only alternatives.
NUZ-001 targets TDP-43 protein aggregation, a pathological hallmark present in approximately 97% of ALS cases, which partly explains why the compound warrants the investment in formulation flexibility to maximise the breadth of patients who can access and sustain treatment.
Study design and data endpoints
The Phase 1 study will generate data across four categories supporting formulation development and the broader clinical and regulatory program:
- Pharmacokinetic data: Measuring how the oral liquid formulation is absorbed and processed compared to tablet administration
- Safety and tolerability data: Assessing adverse events and tolerability profiles under fed and fasted conditions
- Exploratory biomarker data: Supporting mechanistic understanding and future clinical trial design
- Palatability data: Evaluating taste and acceptability factors relevant to patient adherence
This dataset will inform formulation optimisation, regulatory submissions, and commercial positioning of the oral liquid variant.
Development timeline and broader NUZ-001 programme
Study initiation is targeted for Q3 CY2026, subject to completion of remaining operational and site initiation activities. Study completion is anticipated in Q4 CY2026, representing a compressed timeline from initiation to completion within two quarters.
The formulation study runs in parallel with Neurizon’s participation in the Phase 2/3 HEALEY ALS Platform Trial in the United States. The Australian study advances formulation work while the US trial evaluates efficacy, allowing the company to progress multiple aspects of the NUZ-001 development programme simultaneously.
The HEALEY ALS Platform Trial first dosed patients in early 2025, establishing NUZ-001’s operational presence in the US registrational pathway and providing the efficacy data infrastructure that the Australian formulation study is designed to complement.
| Milestone | Target Timing |
|---|---|
| Study initiation | Q3 CY2026 |
| Study completion | Q4 CY2026 |
For investors, the parallel advancement of formulation work and efficacy trials demonstrates capital-efficient development strategy. The relatively straightforward Phase 1 design, completed within two quarters, minimises execution risk whilst generating data that supports both near-term regulatory interactions and longer-term commercial planning.
Management commentary
Executive Chairman Sergio Duchini framed the approval as reflecting Neurizon’s commitment to developing patient-centred therapeutic solutions in ALS.
Sergio Duchini, Executive Chairman
“The oral liquid formulation was developed in direct response to the practical challenges faced by people living with ALS, particularly as swallowing difficulties become more prominent during disease progression. In addition to supporting flexibility and continuity of treatment, the formulation is intended to improve the overall practicality of administration for patients, caregivers and clinical teams.”
Duchini emphasised that advancing the oral liquid formulation reflects Neurizon’s broader strategy to support long-term treatment accessibility and expand the potential utility of NUZ-001 across different stages of disease progression.
The next major ASX story will hit our subscribers first
What this means for Neurizon’s ALS development strategy
The ethics approval demonstrates Neurizon is executing on its stated development roadmap. The company has been advancing development of the oral liquid formulation, and this HREC approval confirms progression from strategic intent to clinical execution within the stated timeframe.
The formulation study expands the potential utility of NUZ-001 across different stages of ALS progression, addressing a practical barrier that limits treatment duration for tablet-only therapies. For a clinical-stage biotechnology company, milestone delivery builds investor confidence in management’s ability to advance programmes toward potential regulatory approval.
NUZ-001 remains an investigational product not approved for commercial use in any jurisdiction. The Phase 1 formulation study forms part of a broader development programme positioning NUZ-001 for clinical, regulatory, and commercial advancement across multiple jurisdictions and patient populations.
Don’t Miss the Next Biotech Breakthrough
Join 20,000+ investors receiving FREE breaking ASX news delivered within minutes of release, complete with in-depth analysis. Click the “Free Alerts” button at Big News Blast to get biotech announcements in your inbox the moment they break, with expert coverage already done.